Extracellular vesicles (EVs) are inherently heterogeneous, meaning that robust analysis requires single-particle molecular characterisation.
To address this need, researchers from the University of Technology Sydney published a paper detailing an improved method for quantitative, single-particle EV phenotyping.
The study, published in November 2025 in the Royal Society of Chemistry publication Sensors & Diagnostics, focuses on enhancing signals obtained from fluorescent nanoparticle tracking analysis (F-NTA)1.
F-NTA involves labelling target particles with fluorescent antibodies. By using a filter to exclude certain frequencies, you can ‘tell’ the camera in NTA to only pay attention to certain particles, allowing you to quantify target particles based on surface marker expression.
A dual-labelling strategy for simultaneously assessing EVs and lipoproteins
Yiting Liu, lead author and PhD candidate from the university’s School of Biomedical Engineering, summarised their dual-labelling strategy:
“EVs were fluorescently labelled with an EV-associated marker (e.g. anti-CD63), while major non-EV contaminants (lipoproteins) in plasma were independently labelled using an anti-ApoB antibody. We used SEC/qEV to remove unbound fluorophores, which enabled the simultaneous quantification of EVs and lipoproteins at the single-particle level in solution, using F-NTA. With this dual-labelling approach, EV purity can be expressed quantitatively as an EV-to-lipoprotein ratio, rather than being inferred indirectly from bulk protein measurements.”
Important signals were being drowned out by background noise
For F-NTA to work as intended, unbound labels need to be removed. Previously, though, this step was not well-established, and the method suffered from a high level of background noise associated with leftover unbound labels.
Previous efforts to address this included a comparison of lipid- and antibody-based labels, as well as exploration of different washing protocols2. In addition, the use of ultracentrifugation to pellet EVs along with unbound fluorescent labels was thought to exacerbate micelle formation and interfere with downstream fluorescent measurements3.
High purification achieved by placing qEV Isolation at the heart of the protocol
Liu and colleagues had prior experience with Izon’s size exclusion chromatography (SEC) qEV isolation platform and noticed how qEV columns offered a particularly good balance between EV yield and purity for plasma EV isolation4. They also noted that others had reported SEC as having the highest efficiency for removing unbound fluorescent labels5.
The group saw an opportunity to reduce F-NTA’s background noise and improve specificity, and incorporated qEV Isolation into the workflow. The use of qEV1 70 nm columns and the Automatic Fraction Collector allowed consistent portions of the sample to be collected at different stages; first to collect purified EVs, and later (using the same qEV column) to separate unbound labels (Figure 1).
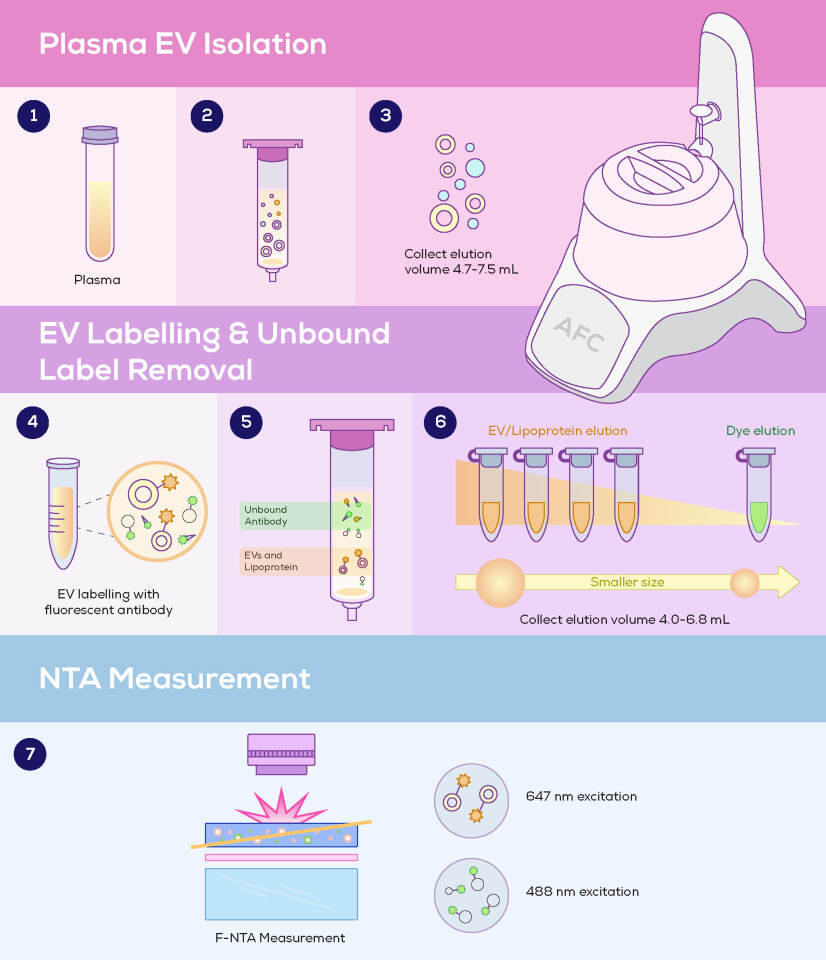
As expected, they found qEV Isolation to be highly effective in removing unbound fluorescent labels. This was critical for reliable F-NTA measurements and enabled them to quantitatively resolve EV subpopulations based on surface marker expression.
Highlights from protocol validation
The group validated the method’s ability to resolve EV subpopulations, and the effectiveness of qEV in removing unbound fluorescent labels. To achieve this, they studied how dual labelling could be used to assess EVs and lipoproteins in parallel and validated this using total internal reflection fluorescence (TIRF) microscopy imaging. Using EVs from different cell lines, they confirmed:
- The effective removal of unbound anti-ApoB labels (AF488 streptavidin) from liposomes. Snapshots from F-NTA visualisation showed a marked reduction in background noise after qEV isolation.
- The versatility of qEV Isolation in removing different labels, as assessed through the quantification of labelled HT29 EVs and very low-density lipoprotein (VLDL) from human plasma. Unremoved labels accounted for 4.22% and 8.66% of total fluorescent particles for EVs and VLDL, respectively. While complete removal of free labels is rarely achievable in fluorescence-based EV workflows, these levels were considered negligible – particularly given that downstream F-NTA analysis applies intensity thresholds to distinguish true particle signals from low-level background.
The research aimed to overcome the challenge of non-specificity, whereby EVs cannot be distinguished from other particle types.
Single-particle analysis key to enhancing EV research and applications
The authors address an important mismatch, where EVs are diverse, and encompass multiple distinct subpopulations – yet bulk analytical methods such as Western blotting and ELISAs lack the resolution that is needed to provide in-depth insights.
“This work extends conventional NTA from a bulk sizing and counting tool into a quantitative single-particle phenotyping platform. It is just like flow cytometry, but much easier to use,” concludes Yiting. “Researchers can now quantify surface marker-positive EV subpopulations directly at the particle level. This provides a practical and accessible method to profile multiplexed nanoparticle populations within a single sample.”
Looking ahead, the authors cite opportunities to enhance the technology’s detection limit and ability to detect rare EV populations. These include the use of novel fluorophores and nanoprobes, increased camera sensitivity, and software updates to enhance analytical capabilities.
As is the case across different fields of nanomedicine, the refinement of single-particle analytical technologies remains a critical way forward.
Learn more about the qEV Isolation platform.









